 Predict which forms an anion, which forms a cation, and the charges of each ion. Many of the nonmetals combine with oxygen to make anions (negatively charged ions) that we are sometimes referred to as oxyanions. One can use the periodic table to predict whether an. The good news, is there are dozens of other polyatomic ions that are relatively easy to figure just by knowing or observing a few patterns. Writing formulas with polyatomic ions follows the same rules as with monatomic ions. (b) A sodium cation (Na+) has lost an electron, so it has one more proton (11) than electrons (10), giving it an overall positive charge, signified by a superscripted plus sign. Take the polyatomic ion section of the periodic chart, copy the formula and the charge in each square from a polyatom ion table from the chemistry textbooks. periodic table tend to form ions having the same charge. Step 3: Determine the charges using the periodic table and the table of polyatomic ions. 1: (a) A sodium atom (Na) has equal numbers of protons and electrons (11) and is uncharged. Note that only two polyatomic ions in this table are cations, hydronium ion (H 3 O +) and ammonium ion (NH 4+ ), the remaining. Step 2: Write the formula of the polyatomic ion. Polyatomic ions have characteristic formulas, names, and charges that should be memorized. The examples include cations like ammonium ion ( NH+4 NH 4 + ), and hydronium ion ( H3O+ H 3 O + ) and anions like hydroxide ion ( OH OH ), and. Because these ions contain more than one atom, they are called polyatomic ions. Polyatomic ions are molecular ions composed of two or more atoms bonded by covalent bonds and acting as a single unit, but unlike molecules, they have a net charge on them. Similarly, we can think of a polyatomic ion as a molecule that has been ionized by gaining or losing electrons. For example, NO3 NO 3 is the nitrate ion it has one nitrogen atom and three oxygen atoms and an overall 1 charge. Some ions consist of groups of atoms bonded together and have an overall electric charge. Since there is one extra electron compared to the number of protons, the ion has a net charge of 1. They contain different numbers of protons and electrons. Periodic Table of Elements ILA Be 9.01 Beryllium Mg 24. 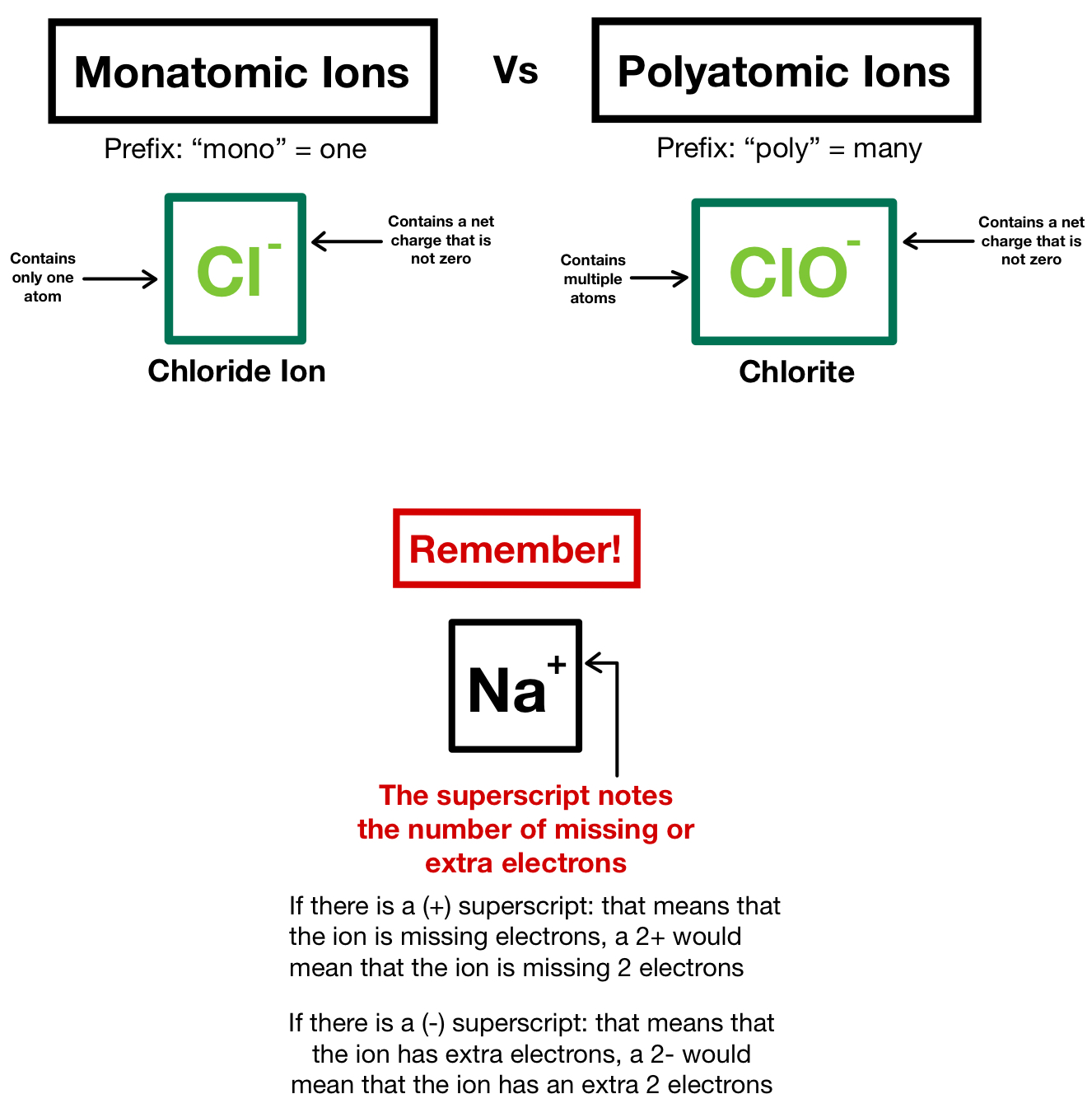
Within a group (family) of elements, atoms form ions of a certain charge.\): Some elements exhibit a regular pattern of ionic charge when they form ions.Īluminum and carbon react to form an ionic compound. 1 lists the ion names and ion formulas of the most common polyatomic ions. How to read the periodic table Periodic table trends Ion-dipole forces Polyatomic ions Ionization energy trend Introduction to Ions, Cations and Anions What is an Ion The definition of an ion is a particle, atom, or molecule with an imbalance of electrical charge. Names and Charges of Polyatomic Ions Transition Metals can have different Oxidation. /PeriodicTableCharge-WBG-56a12db23df78cf772682c37.png)
The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table. Note that only two polyatomic ions in this table are cations, hydronium ion (H 3 O +) and ammonium ion (NH 4 +), the remaining polyatomic ions are all negatively-charged and, therefore, are classified as anions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
